Showing posts from August 17, 2010Show all
Acids, Bases and Buffers
Faizan Bhatti
3:36 PM
Electrolyte is a substrate that conducts electricity when in solution, such as sodium …
Compounds and Molecules as Aggregates of Atoms
Faizan Bhatti
3:23 PM
In addition to being an element, a substance can also be a compound. Compound is compo…
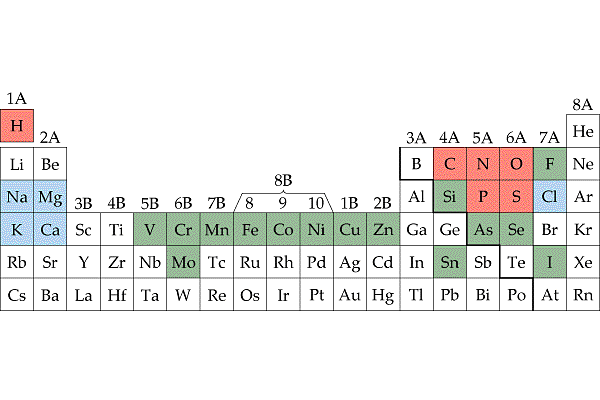
Atoms and Elements as Building Blocks
Faizan Bhatti
3:14 PM
Matter occupies space and has mass. It includes solid, liquids and gases of the enviro…
Popular Posts
Menu Footer Widget
Copyright ©
Notes for Pakistan





Social Plugin